Process FMEA
– Process Failure Mode & Effects Analysis –

Introduction to Process Failure Mode and Effects Analysis (PFMEA)
Manufacturing and Process Engineers envision a process is free of errors. Unfortunately, errors and especially errors propagated when people are present can be quite catastrophic. Process Failure Mode and Effects Analysis (PFMEA) looks at each process step to identify risks and possible errors from many different sources. The sources most often considered are:
- Man
- Methods
- Material
- Machinery
- Measurement
- Mother Earth (Environment)
What is Process Failure Mode and Effects Analysis (PFMEA)
PFMEA is a methodical approach used for identifying risks on process changes. The Process FMEA initially identifies process functions, failure modes their effects on the process. If there are design inputs, or special characteristics, the effect on end user is also included. The severity ranking or danger of the effect is determined for each effect of failure. Then, causes and their mechanisms of the failure mode are identified. The assumption that the design is adequate keeps the focus on the process. A high probability of a cause drives actions to prevent or reduce the impact of the cause on the failure mode. The detection ranking determines the ability of specific tests to confirm the failure mode / causes are eliminated. The PFMEA also tracks improvements through Risk Priority Number (RPN) reductions. By comparing the before and after RPN, a history of improvement and risk mitigation can be chronicled.
Why Perform Process Failure Mode and Effects Analysis (PFMEA)
Risk is the substitute for failure on new processes. It is a good practice to identify risks for each process step as early as possible. The main goal is to identify risk prior to tooling acquisition. Mitigation of the identified risk prior to first article or Production Part Approval Process (PPAP) will validate the expectation of superior process performance.
Risks are identified on new technology and processes, which if left unattended, could result in failure. The PFMEA is applied when:
- There is a new technology or new process introduced
- There is a current process with modifications, which may include changes due to updated processes, continuous Improvement, Kaizen or Cost of Quality (COQ).
- There is a current process exposed to a new environment or change in location (no physical change made to process)
How to Perform Process Failure Mode and Effects Analysis (PFMEA)
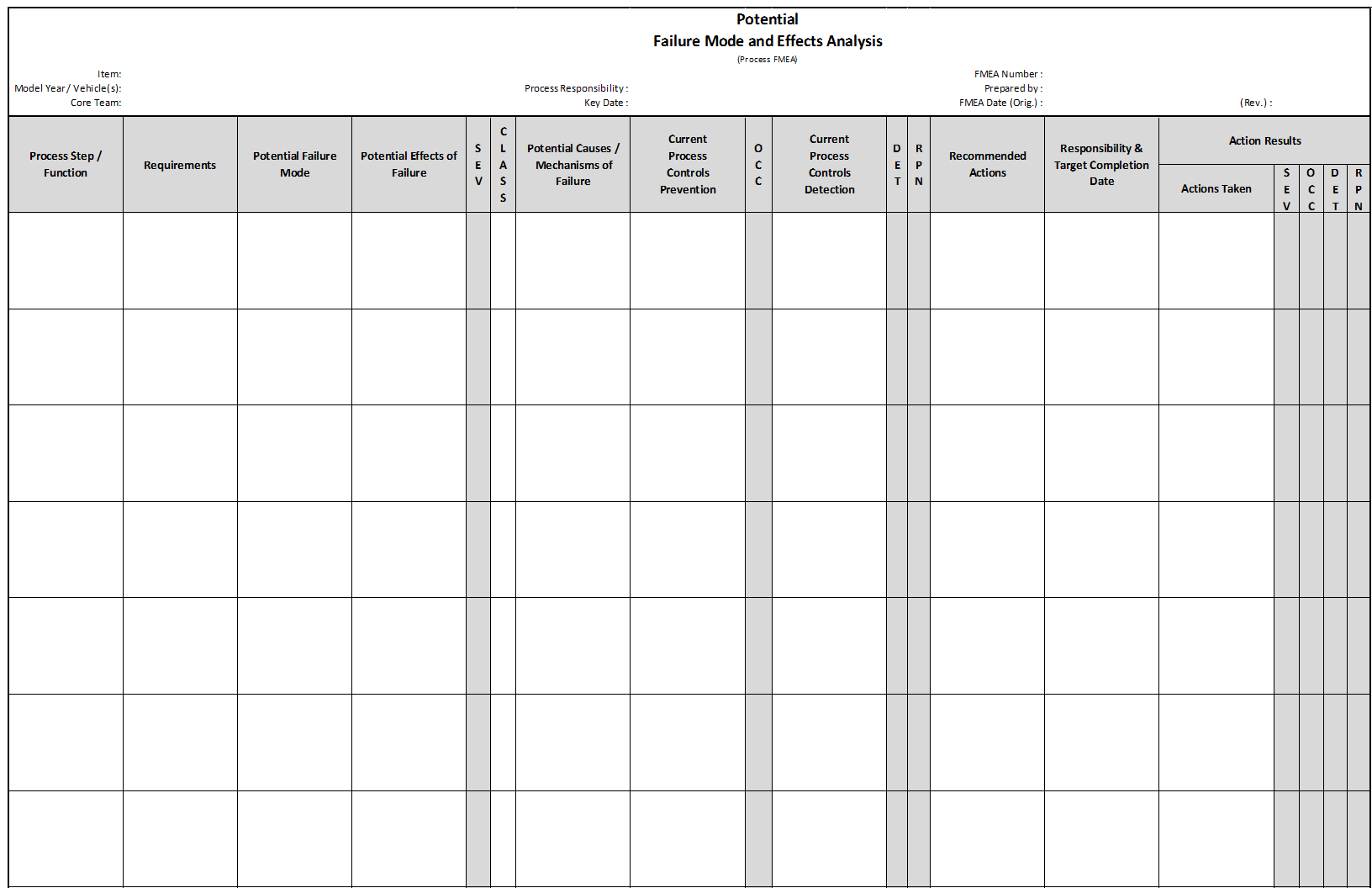
There are five primary sections of the Process FMEA. Each section has a distinct purpose and a different focus. The PFMEA is completed in sections at different times within the project timeline, not all at once. The Process FMEA form is completed in the following sequence:
PFMEA Section 1 (Quality-One Path 1)
Process Name / Function
The Process Name / Function column permits the Process (PE) or Manufacturing Engineer (ME) to describe the process technology that is being analyzed. The process can be a manufacturing operation or an assembly. The function is the “Verb-Noun” that describes what the process operation does. There may be many functions for any one process operation.
Requirement
The requirements, or measurements, of the process function are described in the second column. The requirements are either provided by a drawing or a list of special characteristics. A Characteristics Matrix, which is form of Quality Function Deployment (QFD), may be used and will link characteristics to their process operations. The requirement must be measurable and should have test and inspection methods defined. These methods will later be placed on the Control Plan. The first opportunity for recommended action may be to investigate and clarify the requirements and characteristics of the product with the design team and Design FMEA.
Failure Mode
Failure Modes are the anti-functions or requirements not being met. There are 5 types of Failure Modes:
- Full Failure
- Partial Failure
- Intermittent Failure
- Degraded Failure
- Unintentional Failure
Effects of Failure
The effects of a failure are focused on impacts to the processes, subsequent operations and possibly customer impact. Many effects could be possible for any one failure mode. All effects should appear in the same cell next to the corresponding failure mode. It is also important to note that there may be more than one customer; both internal and external customers may be affected.
Severity
The Severity of each effect is selected based on both Process Effects as well as Design Effects. The severity ranking is typically between 1 through 10.
Typical Severity for Process Effects (when no Special Characteristics / design inputs are given) is as follows:
- 2-4: Minor Disruption with rework / adjustment in stations; slows down production (does not describe a lean operation)
- 5-6: Minor disruption with rework out of station; additional operations required (does not describe a lean operation)
- 7-8: Major disruption, rework and/or scrap is produced; may shutdown lines at customer or internally within the organization
- 9-10: Regulatory and safety of the station is a concern; machine / tool damage or unsafe work conditions
Typical Severity for Design Effects (when Special Characteristics / design inputs are given) is as follows:
- 2-4: Annoyance or squeak and rattle; visual defects which do not affect function
- 5-6: Degradation or loss of a secondary function of the item studied
- 7-8: Degradation or loss of the primary function of the item studied
- 9-10: Regulatory and / or Safety implications
The highest severity is chosen from the many potential effects and placed in the Severity Column. Actions may be identified to can change the design direction on any failure mode with an effect of failure ranked 9 or 10. If a recommended action is identified, it is placed in the Recommended Actions column of the PFMEA.
Classification
Classification refers to the type of characteristics indicated by the risk. Many types of special characteristics exist in different industries. These special characteristics typically require additional work, either design error proofing, process error proofing, process variation reduction (Cpk) or mistake proofing. The Classification column designates where the characteristics may be identified and later transferred to a Control Plan.
PFMEA Section 2 (Quality-One Path 2)
Potential Causes / Mechanisms of Failure
Causes are defined for the Failure Mode and should be determined for their impact on the Failure Mode being analyzed. Causes typically follow the Fishbone / Ishikawa Diagram approach, with the focus of cause brainstorming on the 6M’s: Man, Method, Material, Machine, Measurement and Mother Earth (Environment). Use of words like bad, poor, defective and failed should be avoided as they do not define the cause with enough detail to make risk calculations for mitigation.
Current Process Controls Prevention
The prevention strategy used by a manufacturing or process team may benefit the process by lowering occurrence or probability. The stronger the prevention, the more evidence the potential cause can be eliminated by process design. The use of verified process standards, proven technology (with similar stresses applied), Programmable Logic Controllers (PLC), simulation technology and Standard Work help are typical Prevention Controls.
Occurrence
The Occurrence ranking is an estimate based on known data or lack of it. The Occurrence in Process FMEAs can be related to known / similar technology or new process technology. A modification to the ranking table is suggested based on volumes and specific use.
Typical Occurrence rankings for new process technology (similar to DFMEA Occurrence Ranking) are as follows:
- 1: Prevented causes due to using a known design standard
- 2: Identical or similar design with no history of failure
- This ranking is often used improperly. The stresses in the new application and a sufficient sample of products to gain history are required to select this ranking value.
- 3-4: Isolated failures
- Some confusion may occur when trying to quantify “isolated”
- 5-6: Occasional failures have been experienced in the field or in development / verification testing
- 7-9: New design with no history (based on a current technology)
- 10: New design with no experience with technology
Typical Occurrence rankings for known / similar technology are as follows:
- 1: Prevented through product / process design; error proofed
- 2: 1 in 1,000, 000
- 3: 1in 100,000
- 4: 1 in 10,000
- 5: 1 in 2,000
- 6: 1 in 500
- 7: 1 in 100
- 8: 1 in 50
- 9: 1 in 20
- 10: 1 in 10
Actions may be directed against causes of failure which have a high occurrence. Special attention must be placed on items with Severity 9 or 10. These severity rankings must be examined to assure that due diligence has been satisfied.
PFMEA Section 3 (Quality-One Path 3)
Current Process Controls Detection:
The activities conducted to verify the product meets the specifications detailed by the product or process design are placed in the Current Process Controls Detection column. Examples are:
- Error proofing devices (cannot make nonconforming product)
- Mistake proofing devices (cannot pass nonconforming product)
- Inspection devices which collect variable data
- Alarms for unstable process parameters
- Visual inspection
Detection Rankings
Detection Rankings are assigned to each method or inspection based on the type of technique used. Each detection control is given a detection ranking using a predetermined scale. There is often more than one test / evaluation technique per Cause-Failure Mode combination. Listing all in one cell and applying a detection ranking for each is the best practice. The lowest of the detection rankings is then placed in the detection column. Typical Process Controls Detection Rankings are as follows:
- 1: Error (Cause) has been fully prevented and cannot occur
- 2: Error Detection in-station, will not allow a nonconforming product to be made
- 3: Failure Detection in-station, will not allow nonconforming product to pass
- 4: Failure Detection out of station, will not leave plant / pass through to customer
- 5-6: Variables gage, attribute gages, control charts, etc., requires operator to complete the activity
- 7-8: Visual, tactile or audible inspection
- 9: Lot sample by inspection personnel
- 10: No Controls
Actions may be necessary to improve inspection or evaluation capability. The improvement will address the weakness in the inspection and evaluation strategy. The actions are placed in the Recommended Actions Column.
PFMEA Section 4
Risk Priority Number (RPN)
The Risk Priority Number (RPN) is the product of the three previously selected rankings, Severity * Occurrence * Detection. RPN thresholds must not be used to determine the need for action. RPN thresholds are not permitted mainly due to two factors:
- Poor behavior by design engineers trying to get below the specified threshold
- This behavior does not improve or address risk. There is no RPN value above which an action should be taken or below which a team is excused of one.
- “Relative Risk” is not always represented by RPN
Recommended Actions
The Recommended Actions column is the location within the Process FMEA that all potential improvements are placed. Completed actions are the purpose of the PFMEA. Actions must be detailed enough that it makes sense if it stood alone in a risk register or actions list. Actions are directed against one of the rankings previously assigned. The objectives are as follows:
- Eliminate Failure Modes with a Severity 9 or 10
- Lower Occurrence on Causes by error proofing, reducing variation or mistake proofing
- Lower Detection on specific test improvements
Responsibility and Target Completion Date
Enter the name and date that the action should be completed by. A milestone name can substitute for a date if a timeline shows the linkage between date and selected milestone.
PFMEA Section 5
Actions Taken and Completion Date
List the Actions Taken or reference the test report which indicates the results. The Process FMEA should result in actions which bring higher risks items to an acceptable level of risk. It is important to note that acceptable risk is desirable and mitigation of high risk to lower risk is the primary goal.
Re-Rank RPN
The new (re-ranked) RPN should be compared with the original RPN. A reduction in this value is desirable. Residual risk may still be too high after actions have been taken. If this is the case, a new action line would be developed. This is repeated until an acceptable residual risk has been obtained.
Process Failure Mode and Effects Analysis (PFMEA) Services
The Process FMEA Services available from Quality-One are PFMEA Consulting, PFMEA Training and PFMEA Support, which may include Facilitation, Auditing or Contract Services. Our experienced team of highly trained professionals will provide a customized approach for developing your people and processes based on your unique PFMEA needs. Whether you need Consulting to assist with a plan to deploy PFMEA, Training to help understand and drive improvement or hands-on Project Support for building and implementing your PFMEA process, Quality-One can support you! By utilizing our experienced Subject Matter Experts (SME) to work with your teams, Quality-One can help you realize the value of Process FMEA in your organization.
Learn More About Process Failure Mode and Effects Analysis (PFMEA)
Quality-One offers Quality and Reliability Support for Product and Process Development through Consulting, Training and Project Support. Quality-One provides Knowledge, Guidance and Direction in Quality and Reliability activities, tailored to your unique wants, needs and desires. Let us help you Discover the Value of PFMEA Consulting, PFMEA Training or PFMEA Project Support.